

Water arrives at our reservoirs in the most natural of ways – through precipitation and storm runoff. As water travels over the surface of the land, it dissolves naturally-occurring minerals and can pick up substances resulting from the presence of animals or from human activity.
Contaminants may include: leaves and branches, trash, viruses and bacteria that may come from sewage treatment plants, septic systems, livestock, and wildlife; salts and metals that can be naturally-occurring or result from stormwater run-off, wastewater discharges, and farming; organic chemicals that are by-products of industrial processes and petroleum production, agriculture, gas stations, stormwater run-off, and septic systems and radioactive contaminants, which can be naturally-occurring.
In order to assure that tap water is safe to drink, the Environmental Protection Agency (EPA) sets regulations that limit the amount of certain contaminants in water provided by public water systems. Food and Drug Administration (FDA) regulations set limits for contaminants in bottled water that must provide the same protections for public health.
The Baltimore City Department of Public Works consistently meets or exceeds all federal requirements to provide our customers with the purest, best-tasting water available. How do we do it?
In order to produce water that will meet accepted standards for public drinking water, the following treatment processes are performed: chlorination, coagulation/flocculation, sedimentation, filtration, fluoridation, and pH adjustment
Accepted standards require the finished water to be free of all organisms of a pathogenic nature. In addition, there are limitations on the concentration in the finished water of chemical constituents which are considered harmful or otherwise undesirable. In some localities where raw (non-processed) water has a high mineral content, it is necessary, in addition to the above-mentioned processes, to soften the water.
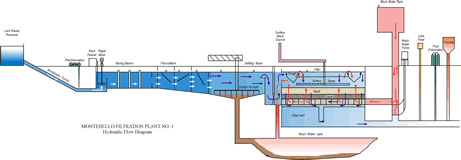
In Baltimore, as in other sections of the country where the mineral content of the raw water is low, the basic treatment procedure is as outlined below for the two Montebello plants and the Ashburton plant.
Baltimore operates three treatment plants that safely produce up to 405 million gallons per day of drinking water. All three treatment plants treat the raw water using the same basic steps: pre-chlorination, coagulation, flocculation, sedimentation, filtration, fluoridation, post-chlorination, and corrosion control treatment. These steps are needed in order to produce water that will meet federal and state drinking water standards, that are free of pathogens, and will be suitable for public consumption.
Chlorine is added to the raw water as it enters each plant. Chlorine kills microorganisms such as bacteria, protozoa, and viruses that may cause illness in humans. Chlorine also prevents the growth of algae at the treatment plant that may interfere with the treatment of water and cause taste and odor problems. Enough chlorine is added to maintain a residual of 1 part per million or residual chlorine in water after filtration. A chlorine residual is needed to prevent re-growth of bacteria in the piping network otherwise known as the distribution system.
 Alum (aluminum sulfate) is added to the water to destabilize natural fine particulate matter suspended in water. This process is known as coagulation. These particles enter the water through land erosion, airborne contaminants, the decay of natural organic matter such as plants and animal wastes, and discharges resulting from human activities. The impurities found in water suspensions consist of charged colloids (colloid - a very small solid suspended in water which varies in size, density, and electric charge) ranging in size from 5 nm to 1 um and particulates greater than 0.5 mm. Most of these particles will not settle by gravity due to their charge and because they are lighter than water (their density is less than water). The addition of alum causes these particles and colloids to clump together to form heavier particles that will settle in water.
Alum (aluminum sulfate) is added to the water to destabilize natural fine particulate matter suspended in water. This process is known as coagulation. These particles enter the water through land erosion, airborne contaminants, the decay of natural organic matter such as plants and animal wastes, and discharges resulting from human activities. The impurities found in water suspensions consist of charged colloids (colloid - a very small solid suspended in water which varies in size, density, and electric charge) ranging in size from 5 nm to 1 um and particulates greater than 0.5 mm. Most of these particles will not settle by gravity due to their charge and because they are lighter than water (their density is less than water). The addition of alum causes these particles and colloids to clump together to form heavier particles that will settle in water.
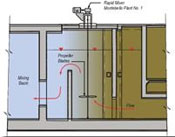
Alum is added quickly to the water at the plants using mechanical mixers. This process is known as Rapid Mix. After passing through the Rapid Mix process, the water travels through flocculation basins where it is gently stirred by slow-moving paddles. Gentle mixing brings small particles into contact so that they will collide, stick together, and grow to a size that will readily settle. These large particles are referred to as floc. The formation of proper floc is necessary for particles to be removed during sedimentation and filtration.
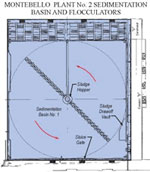 After flocculation, the water and floc move slowly through large basins known as sedimentation or settling basins. The water moves very slowly through these basins due to their large size. This allows the floc to settle to the bottom of the basin. The floc that falls to the bottom of the basins is collected into a hopper by large rotating scrapers where it is removed several times daily by the plant operators. Clearwater above the floc layer (referred to as treatment residuals) flows out of the sedimentation basin and to the filters. Removal of particles in the sedimentation basin improves the operation of the filters that comprise the next treatment process after sedimentation.
After flocculation, the water and floc move slowly through large basins known as sedimentation or settling basins. The water moves very slowly through these basins due to their large size. This allows the floc to settle to the bottom of the basin. The floc that falls to the bottom of the basins is collected into a hopper by large rotating scrapers where it is removed several times daily by the plant operators. Clearwater above the floc layer (referred to as treatment residuals) flows out of the sedimentation basin and to the filters. Removal of particles in the sedimentation basin improves the operation of the filters that comprise the next treatment process after sedimentation.
The sedimentation process removes many particles including clay and silt based turbidity, natural organic matter, and other associated impurities.
These impurities include microbial contaminants, toxic metals, synthetic organic chemicals, iron, manganese, and humic substances. Humic substances come from the soil are produced within natural water and sediments by chemical and biological processes such as the decay of vegetation. Removal of humic substances from drinking water is desirable since they form disinfection byproducts when chlorine is added to the water. At high concentrations, disinfection by-products such as trihalomethanes are a public health concern.
 The last step in purifying the water is accomplished by passing water through a bed of sand and gravel. As water filters through the sand, the remaining particles of suspended matter are trapped in the sand bed. In the filtration process, water flows on top of the sand bed and travels through the bed until it is collected at the bottom in underdrains. Filtered water flows from the underdrains into clear wells or filtered water reservoirs. The rate of filtration is regulated using controllers. The filters must be cleaned periodically as material becomes trapped in the sand and reduces the rate of filtration.
The last step in purifying the water is accomplished by passing water through a bed of sand and gravel. As water filters through the sand, the remaining particles of suspended matter are trapped in the sand bed. In the filtration process, water flows on top of the sand bed and travels through the bed until it is collected at the bottom in underdrains. Filtered water flows from the underdrains into clear wells or filtered water reservoirs. The rate of filtration is regulated using controllers. The filters must be cleaned periodically as material becomes trapped in the sand and reduces the rate of filtration.
Cleaning of the filters is also needed to prevent solids from passing through the filters into the filtered water. To keep the filters operating at their best performance, the filter operators take each filter out of service so they can be back washed. Backwashing cleans the filters by forcing clean water backward through the sand to remove the solids which are deposited in waste drains. Filters are back washed on a rotating schedule to ensure that plants can continuously operate.
Fluoride is added to the filtered water at each of the plants to reduce tooth decay. The plants maintain fluoride levels of approximately 0.7 parts per million in the treated water.
If needed, additional chlorine is added to the finished water that leaves the water plants. Low levels of chlorine (approximately 0.2 to 1.0 part per million) must be maintained in the distribution systems pipes and home plumbing to prevent the growth of microorganisms.
Lime is added to the treated water at the water plants before the water is added to the distribution system. Lime (or calcium oxide) is added to raise the pH of the water to a level of about 8 standard units. The process reduces the ability of the water to corrode water mains and home plumbing materials such as copper, lead, and brass. Corrosive water can dissolve lead and copper. Excessive levels of dissolved lead and copper are a public health concern, particularly for young children.